 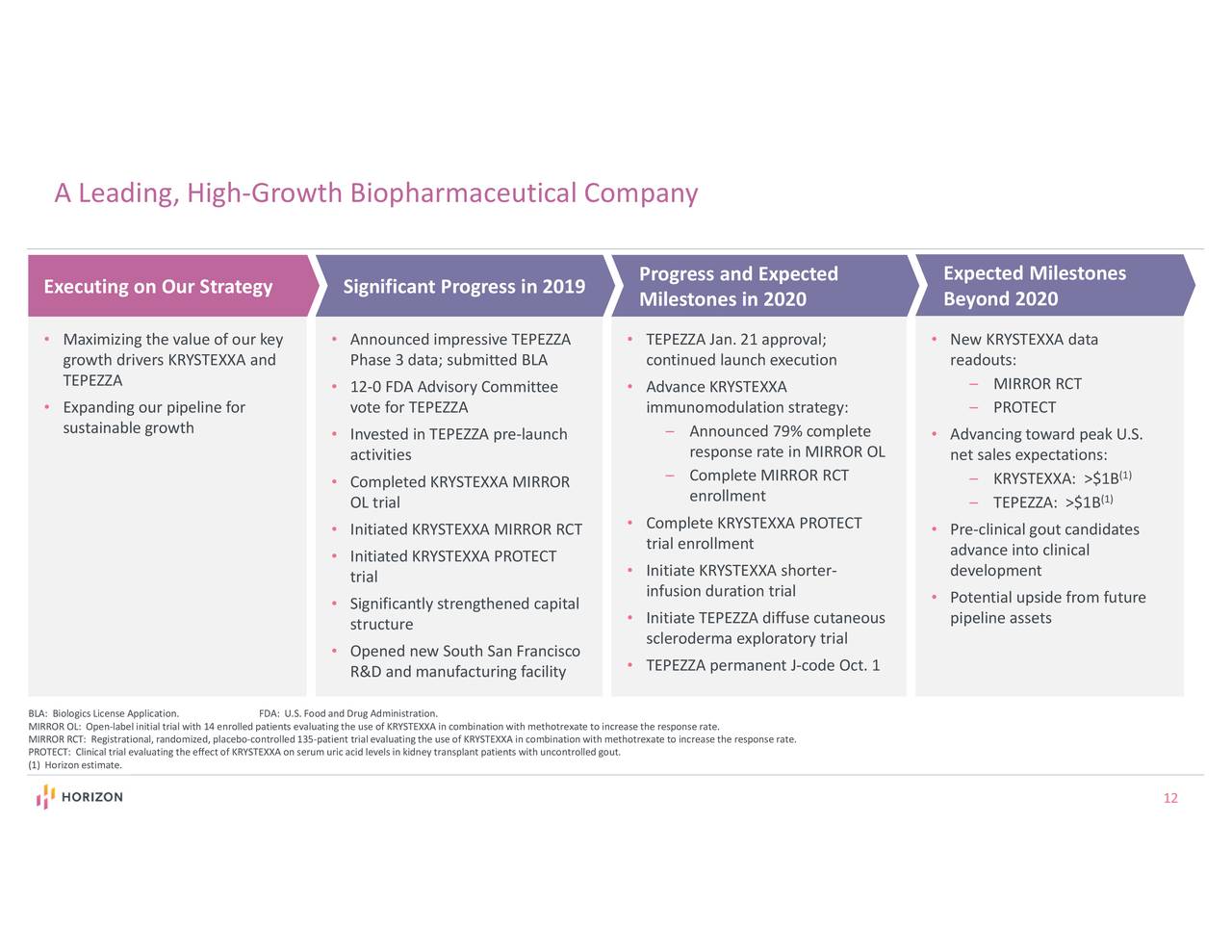
Karin Rosén, M.D., Ph.D., Named Executive Vice President, Research and Development and Chief Scientific Officer. Pursuing TEPEZZA Expansion Outside the United States. Increasing Full-Year 2020 Net Sales Guidance to $2.12 Billion to $2.14 Billion and Full-Year 2020 Adjusted EBITDA Guidance to $920 Million to $940 Million. KRYSTEXXA ® (pegloticase injection) Third-Quarter 2020 Net Sales of $108.5 Million Increasing Full-Year 2020 Net Sales Guidance to Low Double-Digit Growth. TEPEZZA (teprotumumab-trbw) Third-Quarter 2020 Net Sales of $286.9 Million Increasing Full-Year 2020 Guidance to Greater Than $800 Million from Greater Than $650 Million Significantly Increasing Investment in TEPEZZA to Support Continued Strong Growth. Quarterly Orphan Segment Net Sales Increased 131 Percent to $534.8 Million, Representing Nearly 85 Percent of Total Company Net Sales. By the end of the third fiscal year after the merger is complete, the company estimates that the agreement will boost efficiency for the merged group, resulting in an annual pre-tax cost reduction of at least $500 million.- Record Third-Quarter 2020 Net Sales of $636.4 Million Increased 90 Percent Third-Quarter 2020 GAAP Net Income of $292.8 Million Adjusted EBITDA of $329.8 Million. The acquisition of Horizon will drive growth in Amgen's revenue and non-GAAP EPS and is expected to be accretive from 2024."Īmgen has a $28.5 billion credit agreement with Citibank and Bank of America and aims to fund the transaction using debt and cash. Additionally, the potential new medicines in Horizon's pipeline strongly complement our own R&D portfolio. “Amgen's decades of leadership in inflammation and nephrology, combined with our global presence and world-class biologics capabilities, will enable us to reach many more patients with first-in-class medicines like Tepezza, Krstexxa, and Uplinza. 
Bradway, chairman and chief executive officer of Amgen, said in a company announcement. "The acquisition of Horizon is a compelling opportunity for Amgen and one that is consistent with our strategy of delivering long-term growth by providing innovative medicines that address the needs of patients who suffer from serious diseases,” Robert A. Horizon acquired the rights to Upliza after acquiring Viela Bio in 2021 for about $3 billion. The monotherapy drug is the first and only B cell-depleting antibody approved for the disease. 
Uplizna (inebilizumab-cdon), which received FDA approval in June 2020, is developed for the treatment of adults with neuromyelitis optica spectrum disease (NMOSD). Tepezza is Horizon's largest selling drug with sales of $1.66 billion in 2021. Tepezza was the first drug approved for the treatment of TED. The FDA approved Tepezza (teprotumumab-trbw) in January 2020 for the treatment of people with thyroid eye disease (TED), a rare disorder in which the fatty tissues and muscles behind the eyes become inflamed, pushing the eyes forward and outwards.). The deal is expected to close in the first half of 2023. The company will pay $116.50 in cash per share, a premium of 19.7% to Horizon’s last closing price. 
Amgen announced it is acquiring rare disease drug company Horizon Therapeutics, maker of eye disease drugs Tepezza and Uplizna, in a deal valued at $27.8 billion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
